Why this matters: AI is transforming pharmaceutical R&D. Understanding CDER's approach is essential for sponsors, developers, and regulators working at the intersection of AI and drug approval.
🔬 Attention Activity: The AI Drug Development Challenge
Imagine this scenario: A pharmaceutical company uses AI to identify a promising drug candidate, design clinical trials, predict patient responses, and monitor safety signals—all before submitting to FDA.
Quick thought experiment: If you were an FDA reviewer, what would you need to know about an AI model before trusting its predictions for drug approval?
This lesson explores CDER's comprehensive framework for artificial intelligence in drug development—from discovery through post-market surveillance. You'll learn the seven-step credibility process, ten guiding principles, and real-world applications that are reshaping pharmaceutical regulation.
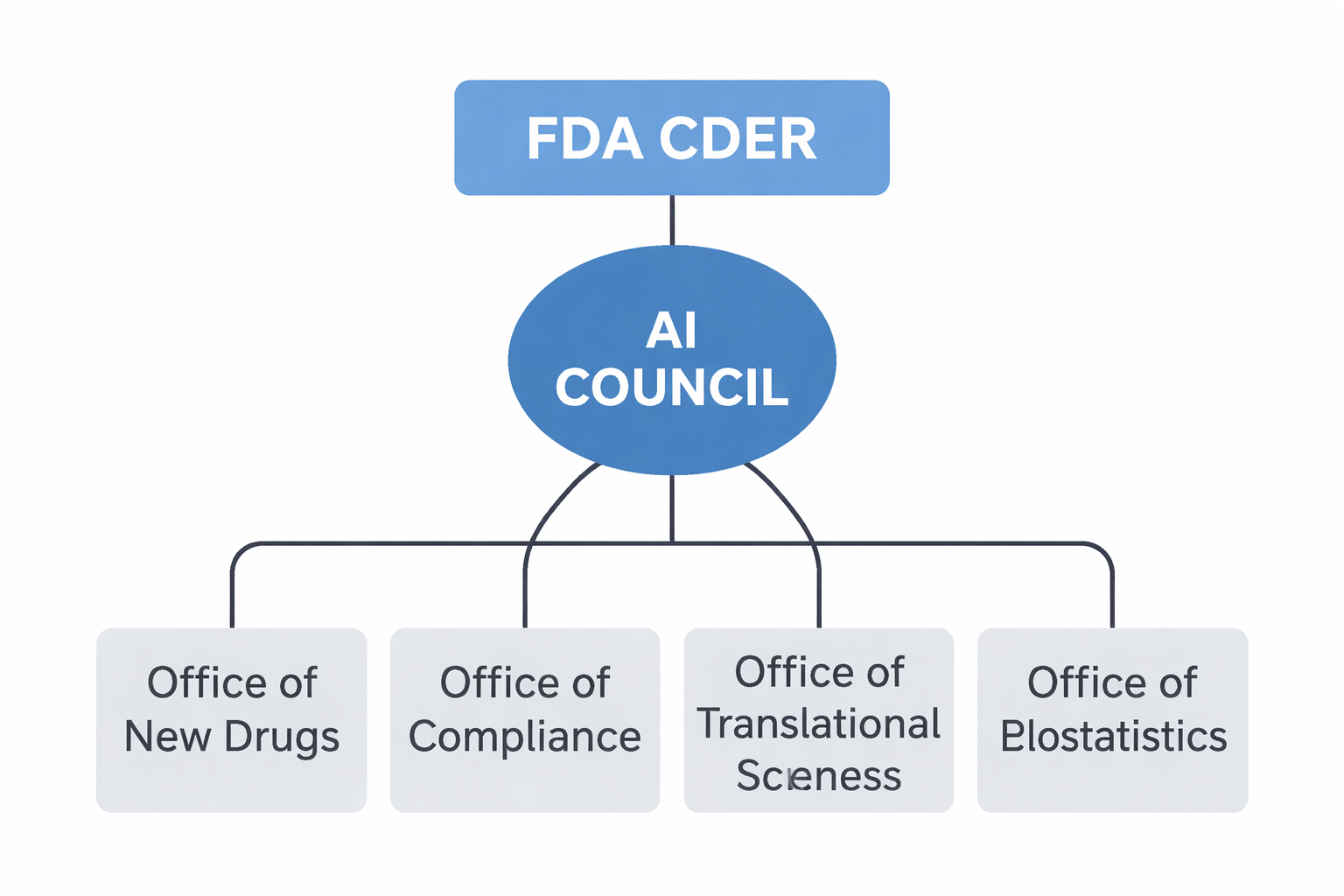
CDER's AI Landscape
The Center for Drug Evaluation and Research (CDER) recognizes AI's transformative potential across the entire drug development continuum.
Why AI Matters to CDER
- Accelerated Discovery: AI reduces time from target identification to candidate selection
- Enhanced Efficiency: Machine learning optimizes clinical trial design and patient recruitment
- Improved Safety: Predictive models identify adverse events earlier
- Personalized Medicine: AI enables precision dosing and patient stratification
AI Applications: Discovery to Market
AI touches every phase of pharmaceutical development. Here are key application areas CDER evaluates:
FDA Draft Guidance (January 2025)
FDA issued landmark draft guidance: "Considerations for the Use of Artificial Intelligence to Support Regulatory Decision-Making for Drugs and Biological Products."
Key Objectives
- Provide a framework for AI model credibility assessment
- Clarify when and how AI outputs can support regulatory submissions
- Establish expectations for transparency, validation, and documentation
- Encourage early engagement between sponsors and FDA
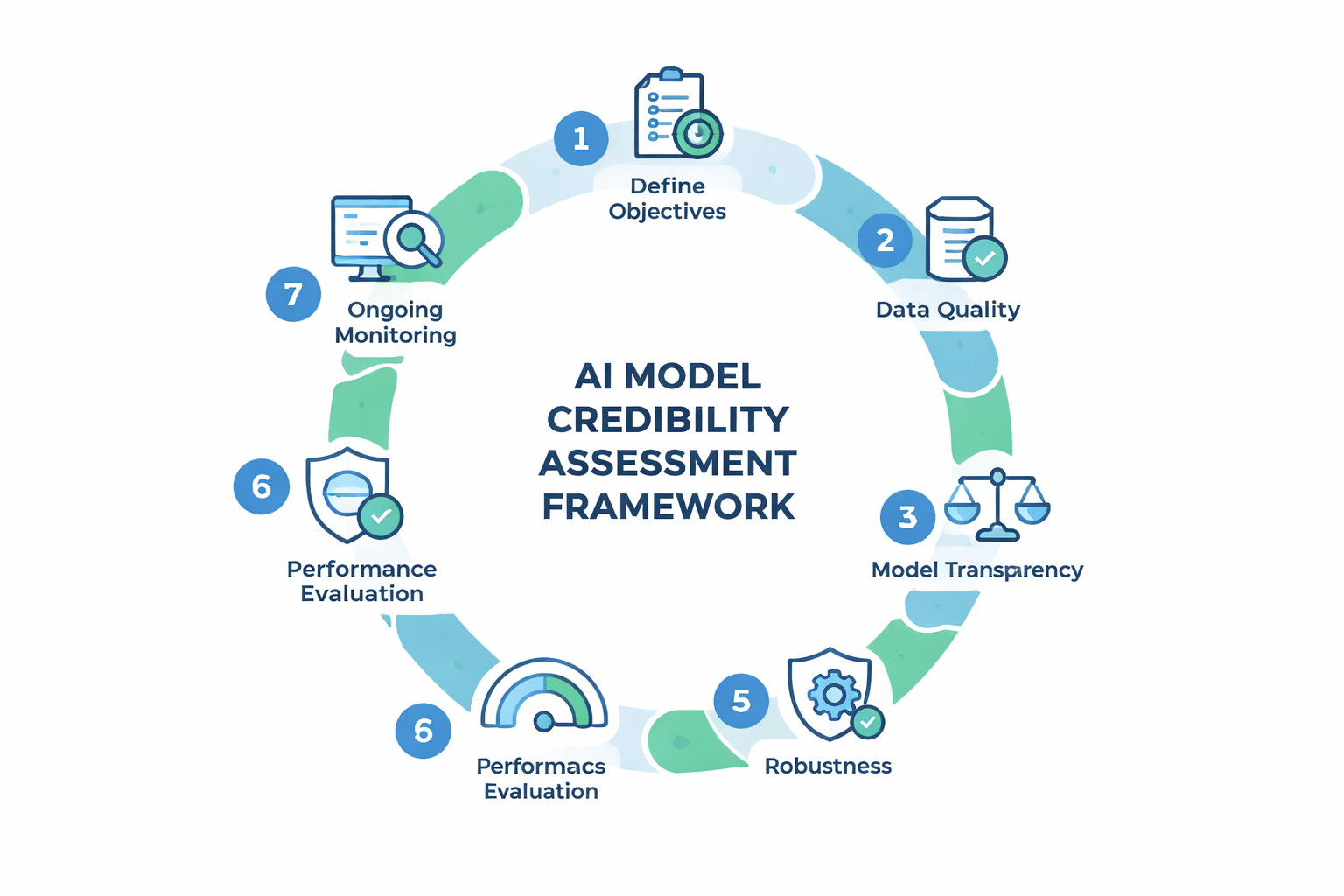
AI Model Credibility Framework
CDER's seven-step process ensures AI models are trustworthy for regulatory decision-making.
Ten Guiding Principles of Good AI Practice
CDER, CBER, and the EMA have established ten core principles to ensure AI/ML tools are safe, effective, and reliable in drug development.
AI must serve human health, with humans retaining final authority in critical safety decisions.
Oversight intensity scales with the stakes: high-impact uses require more rigorous validation.
Compliance with CDISC, ICH, and documented QA/QC processes throughout development.
Predefined scope, limitations, and intended decision points before any model deployment.
Representative, complete, and well-documented training data with clear lineage records.
Open sharing of architecture, training procedures, and performance metrics for verification.
Conducting holdout testing and external validation on independent data before submission.
Post-deployment surveillance to detect model drift and ensure ongoing safety/efficacy.
Identifying and addressing disparities across age, sex, race, and other demographic factors.
Cross-functional teams of clinicians, biostatisticians, and engineers working together.
AI in Clinical Trials & Real-World Evidence
Two critical application areas where CDER sees growing AI adoption:

Clinical Trial Applications
- Patient Recruitment: AI identifies eligible participants from EHRs, reducing enrollment time
- Adaptive Designs: Machine learning enables response-adaptive randomization and dose optimization
- Endpoint Selection: Predictive models identify surrogate endpoints and biomarkers
- Site Selection: Algorithms predict site performance and patient availability
Engaging with CDER on AI
FDA encourages sponsors to engage early when planning to use AI in drug development submissions.
Pre-Submission Meetings
Sponsors should request meetings to discuss:
- AI model credibility assessment strategy
- Data sources and validation approaches
- Context of use and regulatory relevance
- Documentation and transparency plans
Bias, Fairness, and Ethical Considerations
CDER emphasizes algorithmic fairness and bias mitigation to ensure equitable drug development outcomes.
Sources of Bias
- Data Bias: Underrepresentation of demographic groups, health conditions, or geographies in training data
- Algorithmic Bias: Model design choices that amplify disparities
- Deployment Bias: Differential access or interpretation of AI outputs across populations
Mitigation Strategies
AI in Pharmacovigilance
The FDA's Emerging Digital Science and Technology Program (EDSTP) focuses on AI in post-market safety monitoring.
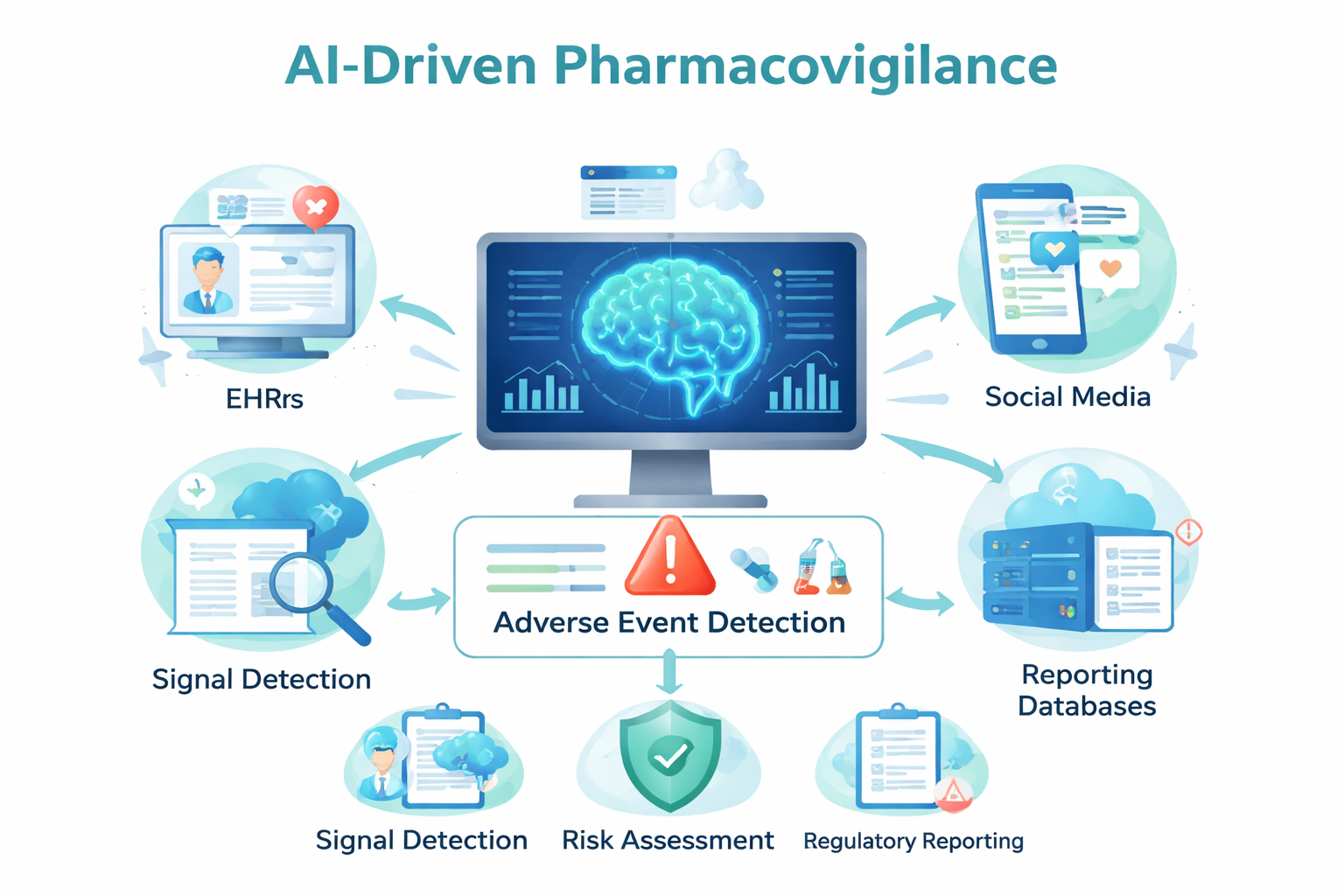
AI Use Cases in Pharmacovigilance
- Signal Detection: AI scans FAERS (FDA Adverse Event Reporting System) for unexpected safety signals
- Social Media Monitoring: Natural language processing extracts adverse event reports from patient forums and social platforms
- EHR Mining: Machine learning identifies patterns in electronic health records
- Risk Prediction: Models predict which patients are at higher risk for specific adverse events
International Regulatory Alignment
CDER recognizes AI is a global challenge requiring harmonized approaches.
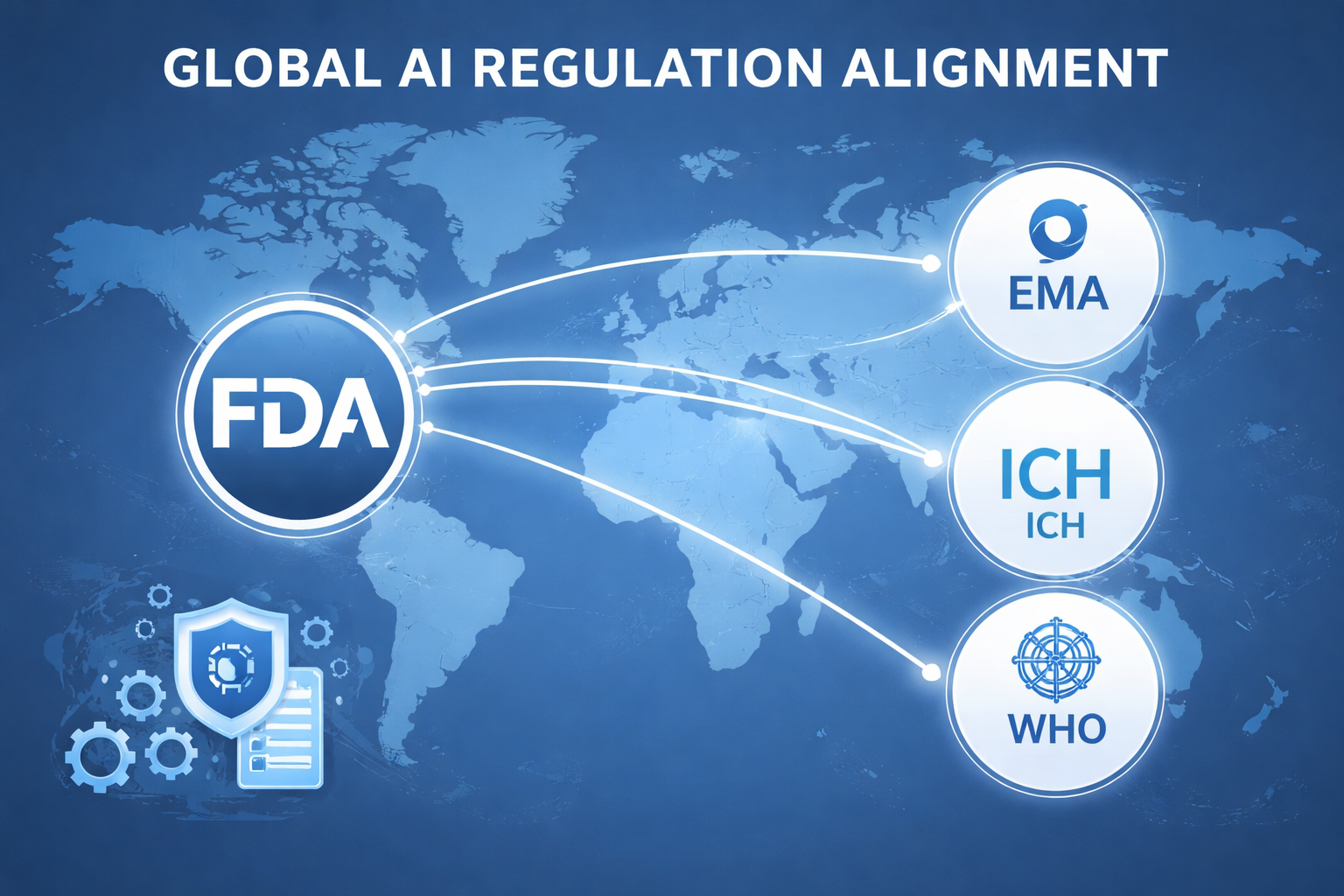
FDA-EMA Collaboration
The ten guiding principles were co-developed with the European Medicines Agency, establishing transatlantic alignment on:
- Human-centric design principles
- Risk-based oversight frameworks
- Data quality and transparency expectations
- Continuous monitoring and lifecycle management
Global AI Initiatives
CDER participates in broader international forums including ICH (International Council for Harmonisation), IMDRF (International Medical Device Regulators Forum), and WHO ethics frameworks for AI in health.
Key Takeaways
Let's consolidate the critical concepts from this lesson:
- CDER AI Council (2024): Provides strategic oversight and coordination of all AI activities within the center.
- Draft Guidance (Jan 2025): Establishes a risk-based framework for AI use in establishing drug safety, effectiveness, and quality.
- Seven-Step Credibility Framework: Define question → Context → Data quality → Development → Validation → Monitoring → Transparency.
- Ten Guiding Principles: Human-centric design, risk-based approach, transparency, fairness, and continuous monitoring are foundational.
- AI Across the Lifecycle: Applications span discovery, preclinical, clinical trials, real-world evidence, and pharmacovigilance.
- Early Engagement: Sponsors should proactively discuss AI strategies with CDER through pre-submission meetings.
- Bias Mitigation: Representative data, subgroup analysis, and algorithmic auditing are regulatory expectations.
- International Alignment: FDA and EMA co-developed principles to harmonize transatlantic AI oversight.
- Transparency & Documentation: Sufficient detail for reproducibility and independent verification is critical.
- Post-Market Flexibility: AI models can evolve post-approval with proper monitoring and FDA notification.
Assessment Time
You've completed the tutorial content. Now it's time to test your understanding of CDER's AI framework.
Instructions:
- You will answer 5 multiple-choice questions
- Each question has 4 options
- Select the best answer for each question
- Your score will be calculated at the end
- You need 80% or higher to earn your certificate
Click Next when you're ready to begin.
